简历:
学习经历
2007~2012四川大学 有机化学专业 理学博士学位,师从冯小明教授从事不对称催化的研究
2003~2007四川大学 应用化学专业 理学学士学位,保送四川大学化学学院有机化学专业攻读博士研究生。
研究及工作经历
2016~至今 四川大学化学学院 教授 博士生导师
2014~2016密西根大学安娜堡分校癌症中心 研究员,合作导师:Dr. Shaomeng Wang
2012~2014 The Scripps Research Institute 研究助理,合作导师:Dr. Philip LoGrasso & Dr. Yangbo Feng
主要研究方向
1. 绿色有机合成方法学(可见光催化或电化学产生的可控自由基反应及其合成应用)
2. 发展小分子催化的高选择性、高效性的自由基(光化学或电化学)参与的不对称合成方法学
3. 小分子药物及靶向抗癌药物的理性设计合成及相关应用
(诚招科研助理、硕博研究生、项目交流生!欢迎本科生同学加入课题组开展“大创”项目和“本科毕业设计”课题!)
主要工作业绩
郑柯博士先后致力于不对称催化剂的设计合成,手性药物合成,神经类药物及抗癌药物和抗癌新靶点等多个前沿性领域的研究,取得了一系列创新性的优秀成果;其丰富的跨学科研究经历,为今后的科研工作,特别是交叉学科和转化药学的研究打下了扎实的基础。回国入职四川大学开始独立工作之后,郑柯博士主要围绕以药物分子为导向的绿色有机合成方法学的研究。重点开展基于利用小分子的弱相互作用来稳定光催化和电催化反应中的高活性中间体,利用产生的高活性的自由基物种来实现以前传统历程不能实现的反应,实现新反应的拓展。同时引入具有手性结构的小分子催化剂来实现一系列自由基参与的不对称催化反应,实现一些以前较难实现的反应,为合成复杂分子和手性药物分子提供一个更绿色、高效的合成方法。最后,我们利用这些方法自身特点和流动化学相结合,为后续大规模的生产创造了便利的条件。至今已在Chem. Rev.,J. Am. Chem. Soc., Angew. Chem. Int. Ed., Nature. Commun.,以及J. Med. Chem.等本领域权威刊物发表SCI论文和专著近40篇,国际专利1篇、美国专利2篇,并多次被Synfacts杂志刊登并给予了高度的评价。
主要奖励和荣誉
2021 四川省学术与技术带头人后备人选
2021四川大学本科优秀毕业论文奖指导教师
2016四川省海外高层次人才引进计划青年项目
2013四川省优秀博士学位论文
2012 “四川省优秀毕业研究生”称号
2010 教育部自然科学奖一等奖(第九完成人)
2010 教育部第一届“博士研究生学术新人奖”
2011 唐敖庆化学奖
2007-2010 曾先后获得四川大学“振兴奖学金”(2007),“陶氏化学教育发展奖学金”(2008),“宝钢优秀学生奖”(2009),“中石化奖学金”(2010)
代表性成果:
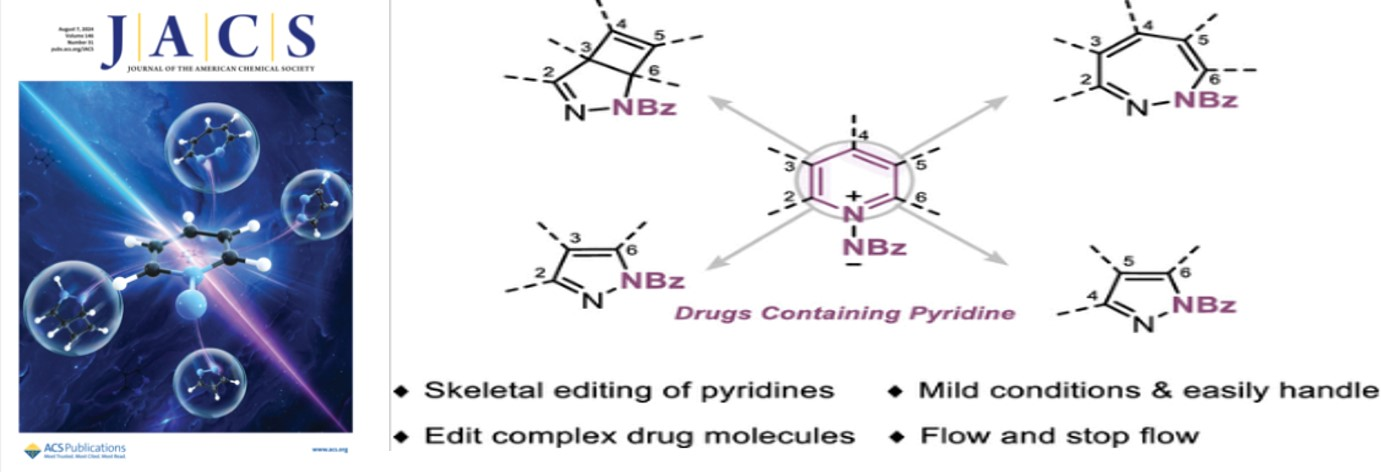
1.Jiajing Luo,§ Qingyang Zhou,§ Zhou Xu, K. N. Houk,* and Ke Zheng*, “Photochemical Skeletal Editing of Pyridines to Bicyclic Pyrazolinesand Pyrazoles” J. Am. Chem. Soc. 2024, doi.org/10.1021/jacs.4c03713 (封面文章)
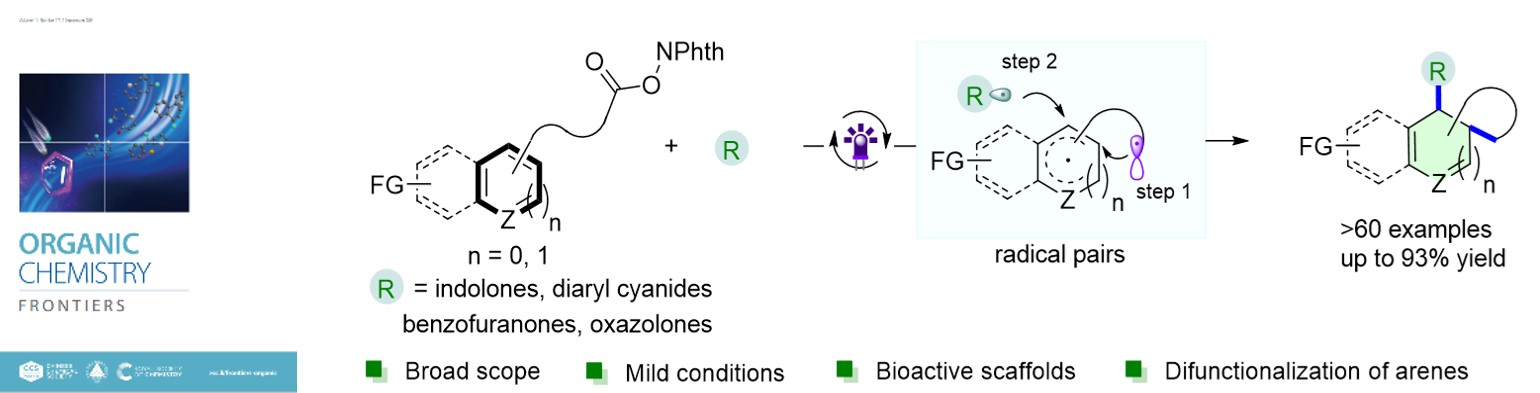
2.Ning Lei, Qian Zhang, Pan Tao, Cong Lu, Qian Lei and Ke Zheng* “Dearomative difunctionalization of arenes via highly selective radical relay reactions” Org. Chem. Front., 2024, DOI: 10.1039/d4qo00964a(封面文章)
3.Ke Zheng, Xiaohua Liu, Xiaoming Feng, “Radical Addition to Carbonyls (or C=X) Enabled by Visible Light Photoredox or Not”, Reference Module in Chemistry, Molecular Sciences and Chemical Engineering, 2024, Elsevier Inc, https://doi.org/10.1016/B978-0-323-96025-0.00042-9

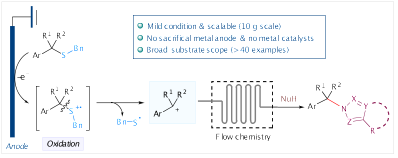
4.Shaopeng Guo,† Yujun Li,† Qing-Han Li* and Ke Zheng* “Electrochemical desulfurative formation of C−N bonds through selective activation of inert C(sp3)-S bonds” Chem. Commun., 2024, 60, 2501–2504
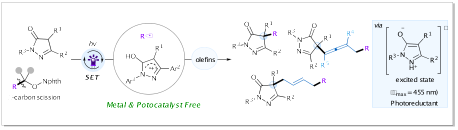
5.Xinxin Geng, Pan Tao, Yujun Li, Ke Zheng* “Photoinduced alkylation of pyrazolones via β-scission of unstrained aliphatic alcohol derivatives” Org. Chem. Front., 2024, 11, 2289–2296
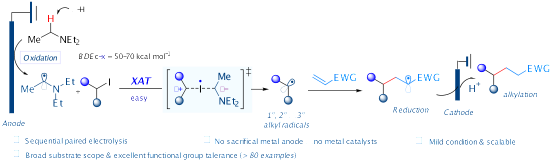
6.Xiang Sun, Ke Zheng* “Electrochemical halogen-atom transfer alkylation via α-aminoalkyl radical activation of alkyl iodides” Nat. Commun. 2023, 14, 6825.
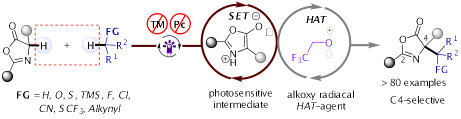
7.Yujun Li, Shaopeng Guo, Qing-Han Li and Ke Zheng* “Metal-free Photoinduced C(sp3)–H/C(sp3)–H Cross-Coupling to access α‑Tertiary Amino Acids Derivatives” Nat. Commun. 2023, 14, 6225.
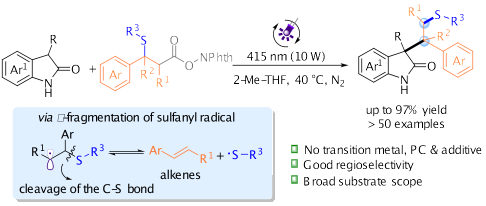
8.Cong Lu, Rui Chen, Rui Wang, Dong Jing and Ke Zheng* “Synthesis of sulfur-containing oxindoles by photoinduced alkene difunctionalization via sulfur 1,2-relocation” Org. Lett. 2023, 25, 5, 750–755
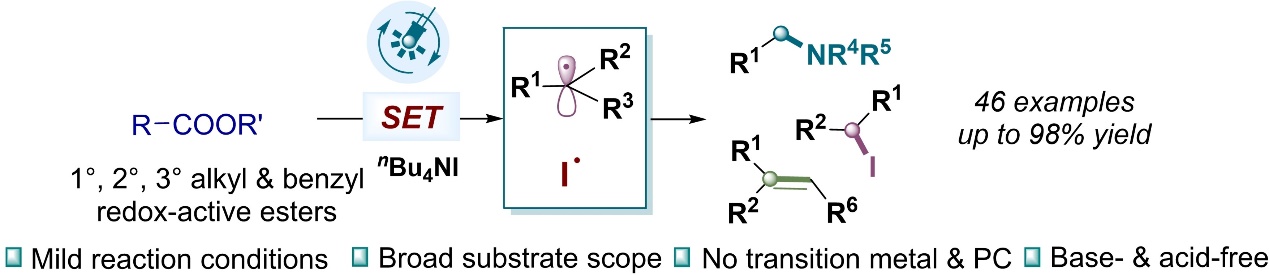
10.Pan Tao,# Yujun Li,# and Ke Zheng* “Electrochemical direct C-H halogenation of N-heteroarenes and naphthols” Eur. J. Org. Chem. 2023, e202300622

11.Yujun Li, Jie Yang, Xinxin Geng, Pan Tao, Yanling Shen, Zhishan Su, and Ke Zheng* “Modular Construction of Unnatural α-Tertiary Amino Acid Derivatives by Multicomponent Radical Cross-Couplings” Angew. Chem. Int. Ed. 2022, 61, e202210755
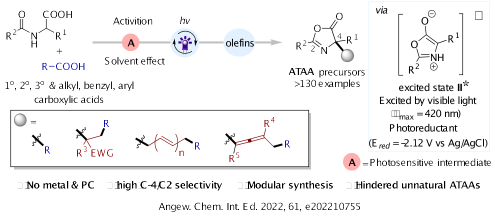
12.Cong Lu, Dong Jing, Yanling Shen, Jiajing Luo and Ke Zheng* “Redox-neutral access to 3,3’-disubstituted oxindoles via radical coupling reactions” Org. Chem. Front., 2022, 9, 4164–4170
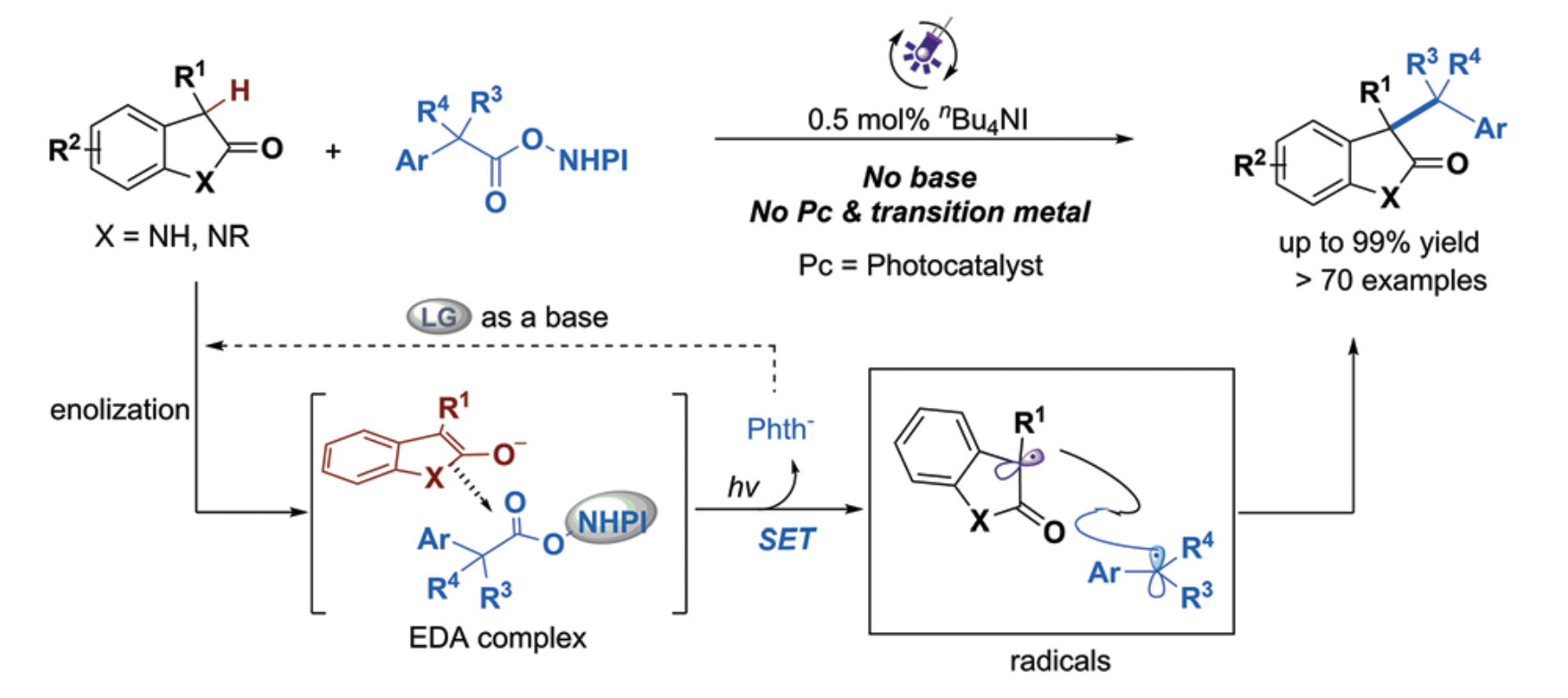
13.Dailin Xi, Cong Lu, Dong Jing, and Ke Zheng* “Carbothiolation of Styrenes by Visible-Light-Induced Thiyl Radicals: C3-Functionalization of Benzofuran-2(3H)-ones“ Eur. J. Org. Chem. 2022, e202200688
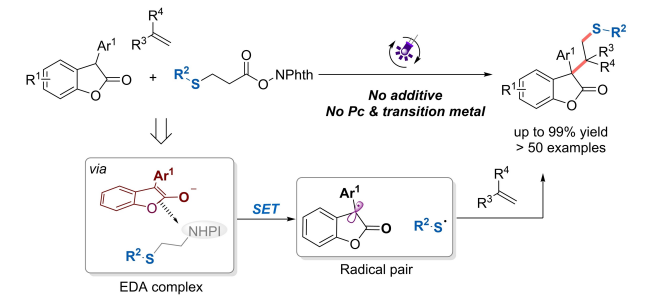
14.Yanling Shen, Ning Lei, Cong Lu, Dailin Xi, Xinxin Geng, Pan Tao Zhishan Su, and Ke Zheng* “Construction of sterically congested oxindole derivatives via visible-light-induced radical-coupling” Chem. Sci., 2021, 12, 15399–15406.

15.Songyang Jin, # Xinxin Geng, # Yujun Li and Ke Zheng* “Phosphoric Acid Mediated Light-Induced Minisci C–H Alkylation of N-Heteroarenes” Eur. J. Org. Chem. 2021, 969–972.
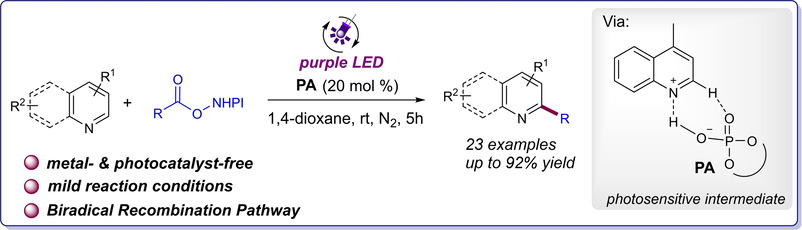
16.Ning Lei, Yanling Shen, Yujun Li, Pan Tao, Liquan Yang, Zhishan Su and Ke Zheng*
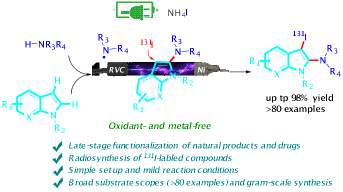
17.“Electrochemical Iodoamination of Indoles using Unactivated Amines” Org. Lett. 2020, 22, 9184–9189
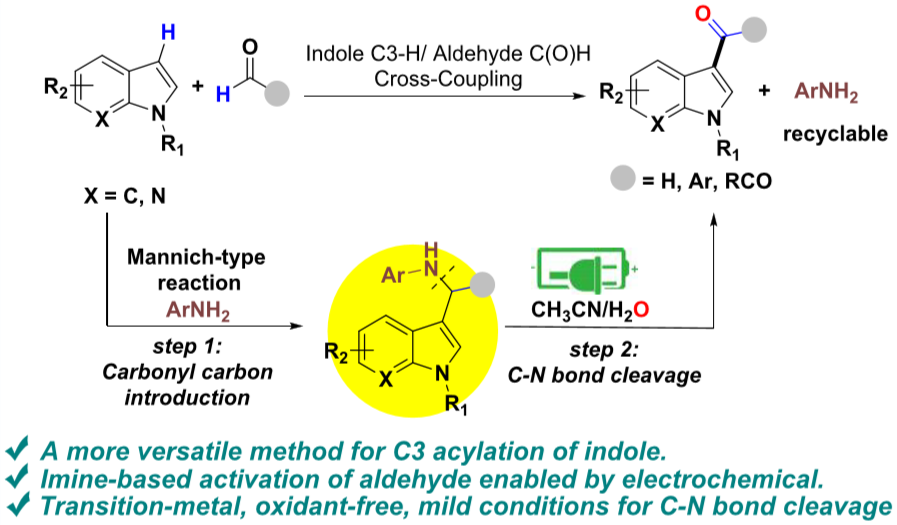
18.Liquan Yang, Zhaoran Liu, Yujun Li, Ning Lei, Yanling Shen, and Ke Zheng* “Electrochemically Enabled C3-Formylation and -Acylation of Indoles with Aldehydes” Org. Lett. 2019, 21, 7702−7707
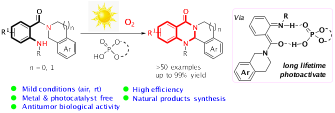
19.Dong Jing,# Cong Lu,# Zhuo Chen, Songyang Jin, Lijuan Xie, Ziyi Meng, Zhishan Su, Ke Zheng* “Light-Driven Intramolecular C-N Cross-Coupling via a Long-Lived Photoactive Photoisomer Complex” Angew. Chem. Int. Ed. 2019, 58, 14666 –14672.
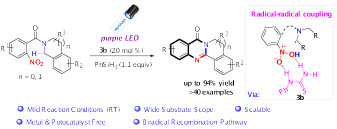
20.Cong Lu# Zhishan Su,# Dong Jing, Songyang Jin, Lijuan Xie, Liangrui, Li and Ke Zheng* “Intramolecular Reductive Cyclization of o-Nitroarenes via Biradical Recombination” Org. Lett. 2019, 21, 1438−1443.

21.Yujun Li, Qi Yang, Liquan Yang, Ning Lei and Ke Zheng* “A scalable electrochemical dehydrogenative cross-coupling of P(O)H compounds with RSH/ROH” Chem. Commun., 2019, 55, 4981-4984.
 A
A
22.ngelo Aguilar, # Ke Zheng, # et al. and Shaomeng Wang * “Structure-Based Discovery of M-89 as a Highly Potent Inhibitor of the Menin-Mixed Lineage Leukemia (Menin-MLL) Protein-Protein Interaction” J. Med. Chem. 2019, 62, 6015−6034.
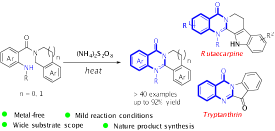
23.Lijuan Xie, Cong Lu, Dong Jing, Xinrui Ou, and Ke Zheng* “Metal-Free Synthesis of Polycyclic Quinazolinones Enabled by a (NH4)2S2O8-Promoted Intramolecular Oxidative Cyclization” Eur. J. Org. Chem. 2019, 3649−3653.
24.Ke Zheng, Xiaohua Liu, and Xiaoming Feng* “Recent Advances in Metal-Catalyzed Asymmetric 1,4-Conjugate Addition (ACA) of Nonorganometallic Nucleophiles” Chem. Rev. 2018, 118, 7586−7656.
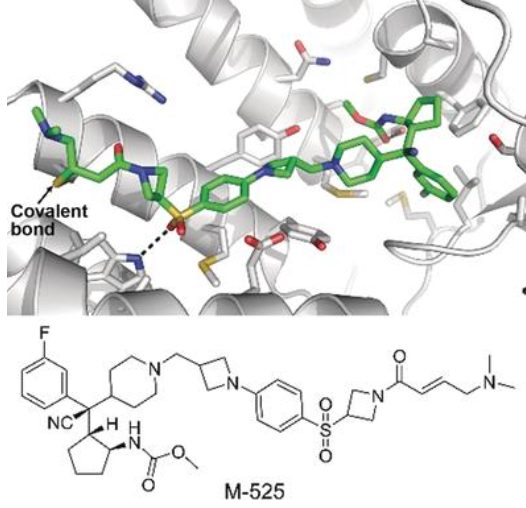
25.Shilin Xu#, Angelo Aguilar#, Tianfeng Xu#, Ke Zheng#, et al. and Shaomeng Wang * “Design of the First-in-Class, Highly Potent Irreversible Inhibitor Targeting the Menin-MLL Protein–Protein Interaction” Angew. Chem. Int. Ed. 2018, 57, 1601−1605.

26.Ke Zheng, Chul Min Park, Sarah Iqbal, Pamela Hernandez, HaJeung Park, Philip V. LoGrasso,* and Yangbo Feng* “Pyridopyrimidinone Derivatives as Potent and Selective c-Jun N-Terminal Kinase (JNK) Inhibitors” ACS Med. Chem. Lett., 2015, 6, 413-418.
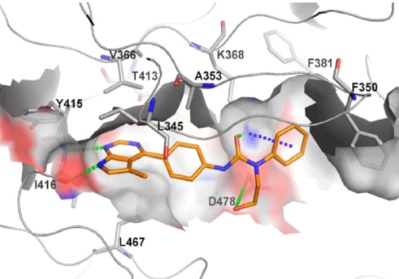
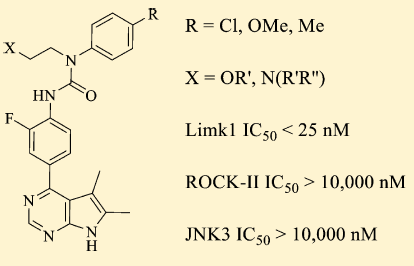
27.Yan Yin, Ke Zheng, Nibal Eid, et al, Philip V. LoGrasso,* and Yangbo Feng*“Bis-aryl Urea Derivatives as Potent and Selective LIM Kinase (Limk) Inhibitors” J. Med. Chem. 2015, 58(4), 1846-1861.

28.Ke Zheng, Sarah Iqbal, Pamela Hernandez, HaJeung Park, Philip V. LoGrasso,* and Yangbo Feng* “Design and Synthesis of Highly Potent and Selective JNK3 Inhibitors: SAR Studies on Aminopyrozal Derivatives” J. Med. Chem. 2014, 57, 10013-10030
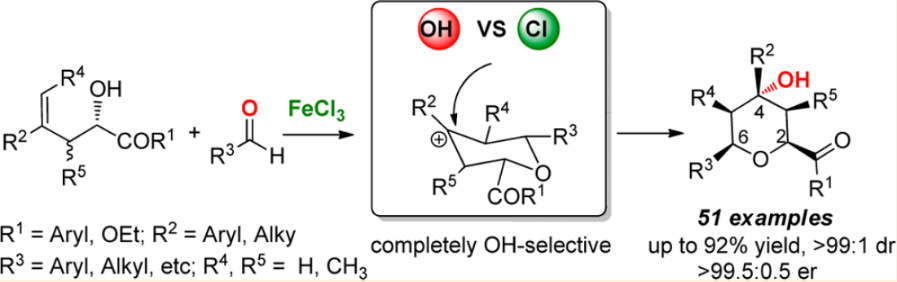
29.Ke Zheng, Xiaohua Liu, Song Qin, Mingsheng Xie, Lili Lin, Changwei Hu, and Xiaoming Feng* “Completely OH-Selective FeCl3 Catalyzed Prins Cyclization: Highly Stereoselective Synthesis of 4-OH-Tetrahydropyrans” J. Am. Chem. Soc. 2012, 134, 17564−17573.

30.Ke Zheng, Lili Lin and Xiaoming Feng* “Chiral N,N '-Dioxide-Ni(II) Complex Catalyzed Asymmetric Carbonyl-Ene Reaction of Ethyl Trifluoropyruvate” Acta Chim. Sinica 2012, 70, 1785-1790.
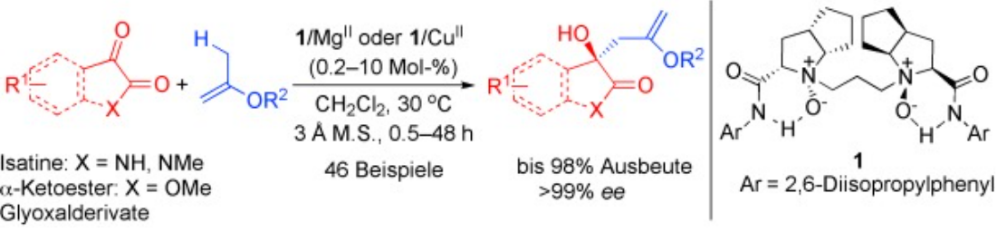
31.Ke Zheng, Chengkai Yin, Xiaohua Liu, and Xiaoming Feng* “Catalytic Asymmetric Addition of Alkyl Enol Ethers to 1,2-Dicarbonyl Compounds: Highly Enantioselective Synthesis of Substituted 3-Alkyl-3-Hydroxyoxindoles” Angew. Chem. Int. Ed. 2011, 50, 2573.
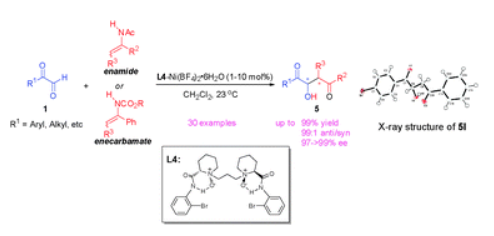
32.Ke Zheng, Xiaohua Liu, Jiannan Zhao, Yang Yang, Lili Lin and Xiaoming Feng* “Highly Enantioselective Aza-Ene-Type Reaction Catalyzed by Chiral N,N’-dioxide-nickel(II) Complex” Chem. Commun. 2010, 46, 3771–3773.
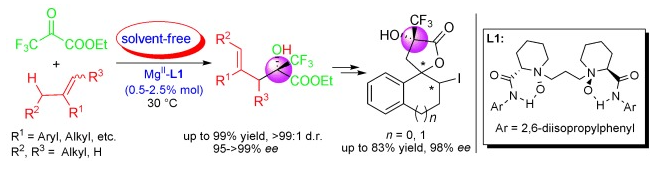
33.Ke Zheng, Yang Yang, Jiannan Zhao, Chengkai Yin, Lili Lin, Xiaohua Liu and Xiaoming Feng*. “Magnesium(II)-Catalyzed Asymmetric Ketone-Ene Reaction: Stereocontrolled Access to Enantioenriched Trifluoromethyl-Substituted Compounds” Chem. Eur. J. 2010, 16, 9969.
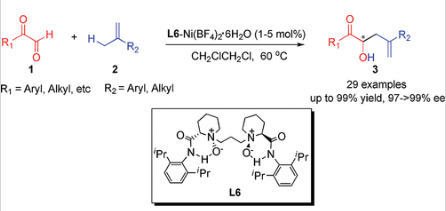
34.Ke Zheng, Jian Shi, Xiaohua Liu, and Xiaoming Feng*. “Asymmetric Carbonyl-Ene Reaction Catalyzed by Chiral N,N’-Dioxide -Nickel(II) Complex: Remarkably Broad Substrate Scope” J. Am. Chem. Soc. 2008, 130, 15770-15771.
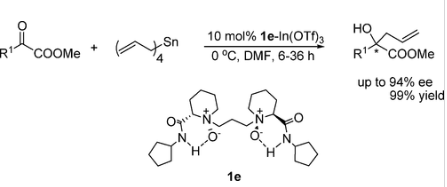
35.Ke Zheng, Bo Qin, Xiaohua Liu and Xiaoming Feng* “Highly Enantioselective Allylation of a-Ketoesters Catalyzed by N,N’-Dioxide-In(III) Complexes” J. Org. Chem. 2007, 72, 8478-8483.
36.Y. Yan, K. Zheng, et al, Y. Feng* and P. LoGrasso*. “Bis-aryl Urea Derivatives as Potent and Selective LIM Kinase (Limk) Inhibitors” J. Med. Chem. 2015, 58(4), 1846-1861.
37.X. Liu, K. Zheng, and X. M. Feng. “Advancements in the Catalytic Asymmetric Intermolecular Ene-Type Reactions” SYNTHESIS, 2014; 46: 2241-2257
38.J. N. Zhao, K. Zheng, et al and X. M. Feng. “Asymmetric Mukaiyama Aldol Reaction Catalyzed by C2-Symmetric N,N’-Dioxide–Ni(II) Complex” Synlett, 2011, 903-906.
39.P. HaJeung; I. Sarah; H. Pamela; M. Rudy; K. Zheng, et al, Y. Feng and P. LoGrasso. “Structural basis and biological consequences for JNK2/3 isoform selective aminopyrazoles” Scientific reports 2015, 5, 8047
40.K. Shen, X. H. Liu, K. Zheng, et al and X. M. Feng. “Catalytic Asymmetric Synthesis of 3-(a-Hydroxy-b-carbonyl) Oxindoles by a Sc(III)-Catalyzed Direct Aldol-Type Reaction” Chem. Eur. J. 2010, 16, 3736-3742.
41.J. Shi, M. Wang, L. He, K. Zheng, et al and X. M. Feng. “Enantioselective Michael addition of malononitrile to chalcones catalyzed by a simple quinine-Al(OiPr)3 complex: a simple method for thesynthesis of a chiral 4H-pyran derivative” Chem. Commun. 2009, 45, 4711–4713.
42.B. Qin, X. H. Liu, J. Shi, K. Zheng, et al and X. M. Feng. “Enantioselective Cyanosilylation of a,a-Dialkoxy Ketones Catalyzed by Proline-Derived in-Situ-Prepared N-Oxide as Bifunctional Organocatalys” J. Org. Chem. 2007, 72, 2374.
